When I was eight or nine year old, I went to the 1963-64 World’s Fair in New York. Among the attractions, in “the kitchen of the future”, I saw the first version of an amazing fry-pan that was coated with plastic. You could cook an egg on that plastic without any oil, and the egg didn’t stick. The plastic was called teflon, a DuPont innovation, whose molecule is shown below.
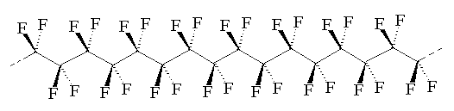
Years later, I came to understand that Teflon’s high-temperature stability and non-stick properties derive from the carbon-fluorine bonds. These bonds are much stronger than the carbon-hydrogen bonds found in food, and most solid, organic things. Because of the strength of the carbon-fluorine bond, Teflon is resistant to oxidation, and to chemical interaction with other molecules, e.g. in food. It does not even interact with water, making it hydrophobic and non-wetting on metals. The carbon-carbon bonds in the middle remained high temperature stable, in part because they were completely shielded by the fluorine atoms.
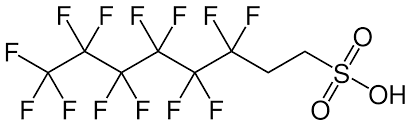
But as remarkable as teflon’s non-stick properties are, perhaps the most amazing thing was that it somehow sticks to the pan. For the first generation pans I saw, it didn’t stick very well. Still, the DuPont engineers had found a way to stick non-stick Teflon to a metal for long enough to cook many meals. If they had not found this trick, teflon would not have the majority of its value, but how did they do it? It turns out they used a thin coating of a di-functional compound called PFAS, a a polyfluoro sulphonyl (or polyfluoroalkyl) substance. The molecular structure of a common PFAS, is shown above.
Each molecule of PFAS has one end that’s teflon-like and another end that’s different. The non-Teflon end, in this case a sulfonyl group, is chosen to be both high temperature stable and sticky to metal oxides. The sulphonyl group above is highly polar, and acidic. Acidic will bind to bases, like metal oxides. The surface of the metal pan is prepared by applying a thin layer of oxide or amidine, making it a polar base. The PFAS is then applied, then Teflon. The Teflon-end of the PFAS is bound to teflon by the hydrophobicity of everything else rejecting it.
There are many other uses for PFAS. For example, PFAS is applied to clothing to make it wrinkle free and stain resistant. It can also be used as a super soap, making uncommonly stable foams and bubbles. It is also used in fire-fighting and plane de-icing. Finally, PFAS is the main component of Nafion, the most common membrane for PEM fuel cells. (I can think of yet other applications..) There is just one small problem with PFAS, though. Like teflon, this molecule is uncommonly stable. It doesn’t readily decompose in nature. That would be a small problem if we were sure that PFAS was safe. As it happens it seems safe, but we’re not totally sure.
The safety of PFAS was studied extensively before PFAS-teflon pans was put on the market, but the methodology has been questioned. Large doses of PFAS were fed to test animals, and their health observed. Since the test animals showed no real signs of ill-health though some showed a slight liver enlargement, PFAS was accepted as safe for humans at a lower exposure dose. PFAS was approved for use on pans and allowed to be dumped under conditions where humans would be exposed to 1/1000 of that used on the animals. The assumption was that there would be little or no health hazard at these low exposure levels.
But low risk is not no risk, and today one can sue for even the hint of an effect though use of a class action suit. That is, lawyers sue on behalf of all the people who might have been damaged. My city was sued successfully this way for complicity in sewage over-flows. Of course, since the citizens being paid by the suit are the same ones who have to pay for the damage, only the lawyers benefit. Still, the law is the law, and at least for some judges, putting anyone at risk is enough evidence of willful disregard to hand down a stinging judgement against the evil doer. Judges have begun awarding large claims for PFAS too. While no individual can get the claim more than a tiny amount of money, the lawyers can do very well.
There is no new evidence that PFAS is dangerous, but none is needed if you can get yourself the right judge. In this regard, an industry of judicial tourism has sprung up, where class-action lawyers travel to districts where the judges are favorable. For Teflon suits, the bust hunting grounds are in New York, New Hampshire, and California, and the worst are blood-red states like Wyoming and Utah. Just as different judges promote different precedents, different states allow vastly different PFAS concentrations in the water. A common standard, one used by Michigan, is 70 ppt, 1 billion times stricter than the amounts tested on animals. This is roughly 500 times stricter than the acceptable concentratios for lead, a known poison. The standard in New York is 7 times stricter than Michigan, 10 ppt. The standard in North Carolina is 140,000 ppt, in in several states there is no legal limit to PFAS dumping. There is no scientific logic to all of this, and skeptical view is that the states that rule more strictly for PFAS than lead do so make money for lawyers. Lead is everyone in the natural environment, so you can’t sue as easily for lead. PFAS is a man-made intruder, though, and a strict standard helps lawyers sue. You can find a summary of state by state regulations here.
Any guideline stricter than about 1000 ppt, presents a challenge to the water commissioner who must measure it and enforce the law. There are tricks, though. You can use the surfactant quality of PFAS to concentrate it by a factor of 100 or more. To do this, you take a sample of river water and create bubbles. Any bubbles that form will be highly concentrated in PFAS. Once PFAS can be identified this way, and the concentrators estimated, the polluters can be held liable. Whether we benefit from the strict rulings is another story. If I were making the law for Michigan, I’d probably choose a limit about 1 ppb, but I’m not making the law. The law, as written, may be an idiot, as Bumble said, but the Law is the Law.
In terms of Michigan fishing, while some rivers have PFAS concentrators above the MI-legal limit, they are generally not far over the line. I would trust the fish in the Huron River, even west of Wixom road but I’d suggest you avoid any foam you find floating there. The PFAS content of foam will be much higher than that of the water in general.
Robert E. Buxbaum, June 30, 2020, edited July 8, 2020. There are seven compounds known as PFAS’s: perfluorooctanesulfonic acid (PFOS), perfluorooctanoic acid (PFOA), perfluorononanoic acid (PFNA), perfluorohexanesulfonic acid (PFHxS), perfluoroheptanoic acid (PFHpA), and perfluorobutanesulfonic acid (PFBS).